44 h2y acid or base
Acid vs Base - Difference and Comparison | Diffen Bases are the chemical opposite of acids. Acids are defined as compounds that donate a hydrogen ion (H +) to another compound (called a base).Traditionally, an acid (from the Latin acidus or acere meaning sour) was any chemical compound that, when dissolved in water, gives a solution with a hydrogen ion activity greater than in pure water, i.e. a pH less than 7.0. Inorganic Exam 2 Flashcards | Quizlet Consider the acid‑base reaction and classify each of the reactants and products as an acid or base according to the Brønsted theory. acid: CF3COOH, H3O+ base: H2O, CF3COO- Consider three generic acids: HX, HY, and HZ. Arrange the acids according to strength. Strongest HX HY HZ Weakest
Chem 121 Exam 3 Flashcards | Quizlet H2Y-: base H2Z-: acid H3Y: acid HZ2-: base Identify the conjugate acid for each base. - conjugate acid of HSO−4: - conjugate acid of SO−4^2- - conjugate acid of NH3 H2SO4 HSO4- NH4+ Identify the pair of species that is not a conjugate acid-base pair. - HNO3; NO−3 - H2SO3; HSO−3 - CH3NH+3; CH3NH2 - H2S; S2− H2S and S2-

H2y acid or base
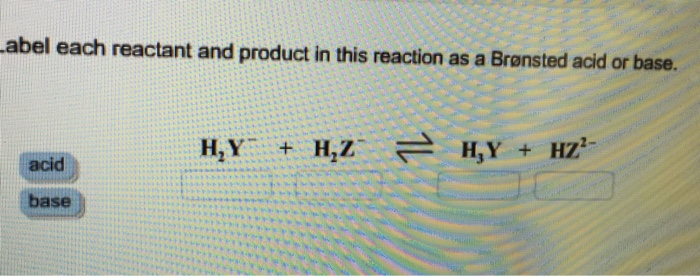
Chem 106 - Sapling Questions - Topic 3 Flashcards | Quizlet Label each reactant and product in this reaction as a Brønsted acid or base. H2Y- + H2Z- <-> H3Y + H3-2 Conjugate pairs H2Y- base-acid H3Y H2Z- acid-base H3-2 conjugate acid of a base is formed by adding a proton (ion) to the base conjugate base of an acid is formed by removing a proton from the acid. Chem 1412 Hw 10 Flashcards | Quizlet Consider three generic acids with the following relative strengths: HX > HY > HZ Rank the strengths of their conjugate bases. Strongest Base Z- Y- X- Weakest Base 6. Complete these Bronsted-Lowry reactions. HS^- + H^+ = H2S HS^- + OH^- = HO+S^2- 7. Rank the oxoacids of Iodine according to strength. Strongest Acid HIO4 HIO3 HIO2 HIO 8. Acid and Base Chart — Table of Acids & Bases - Sigma-Aldrich Acid-Base Pairs and Buffer Chemistry A buffer solution contains a weak acid and its conjugate base or a weak base and its conjugate acid. Buffers work by reacting with a base or acid to control the pH of a solution. Download the Acid & Base Chart Acid & Base Chart Image Acid & Base Chart PDF
H2y acid or base. pH Scale: Acids, bases, pH and buffers (article) | Khan Academy H + ^+ + start superscript, plus, end superscript concentration shifts away from neutral when an acid or base is added to an aqueous (water-based) solution. For our purposes, an acid is a substance that increases the concentration of hydrogen ions (H + ^+ + start superscript, plus, end superscript) in a solution, usually by donating one of its hydrogen atoms through dissociation. Label each reactant and product in this reaction as a Bronsted acid or ... Strong acid Weak acid H2C03 H2SO4 HI HF HNO3 Label each reactant and product in this reaction as a Bronsted acid or base. Q: (16-4) Label each reactant and product in this reaction as a Bronsted acid or base. Q: Label each reactant and product in this reaction as a Bronsted acid or base. H2Y- + H2Z- <===> H3Y + HZ2- Q: Acid and Base Chart — Table of Acids & Bases - Sigma-Aldrich Acid-Base Pairs and Buffer Chemistry A buffer solution contains a weak acid and its conjugate base or a weak base and its conjugate acid. Buffers work by reacting with a base or acid to control the pH of a solution. Download the Acid & Base Chart Acid & Base Chart Image Acid & Base Chart PDF Chem 1412 Hw 10 Flashcards | Quizlet Consider three generic acids with the following relative strengths: HX > HY > HZ Rank the strengths of their conjugate bases. Strongest Base Z- Y- X- Weakest Base 6. Complete these Bronsted-Lowry reactions. HS^- + H^+ = H2S HS^- + OH^- = HO+S^2- 7. Rank the oxoacids of Iodine according to strength. Strongest Acid HIO4 HIO3 HIO2 HIO 8.
Chem 106 - Sapling Questions - Topic 3 Flashcards | Quizlet Label each reactant and product in this reaction as a Brønsted acid or base. H2Y- + H2Z- <-> H3Y + H3-2 Conjugate pairs H2Y- base-acid H3Y H2Z- acid-base H3-2 conjugate acid of a base is formed by adding a proton (ion) to the base conjugate base of an acid is formed by removing a proton from the acid.

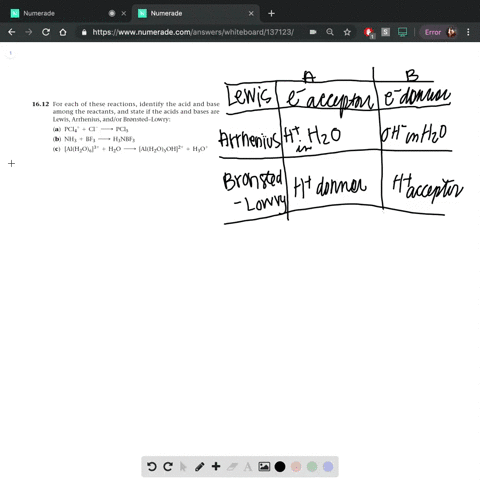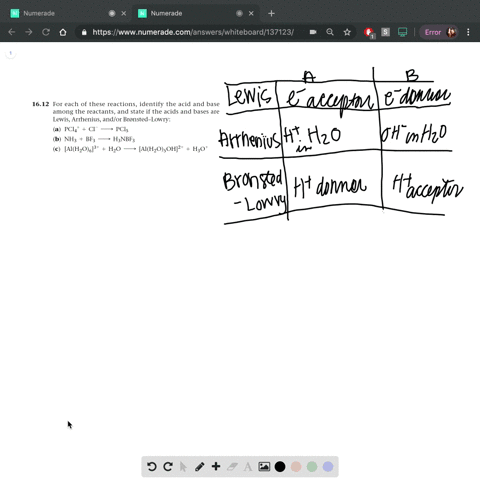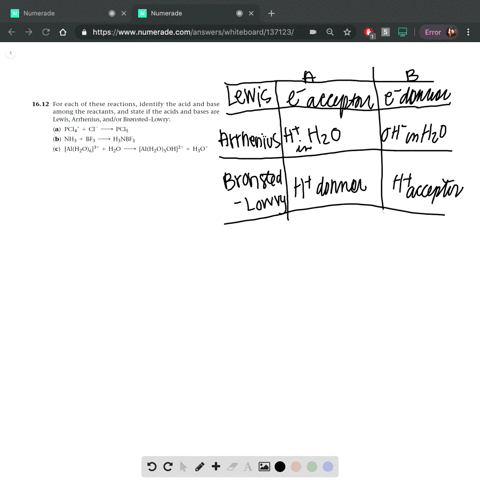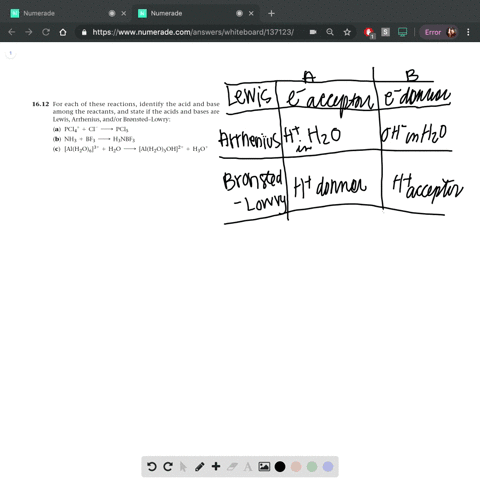
Identify each reactant and product in this reaction as a Bronsted acid or base., Hz Y- + HzZ acid base, H; Y + HZ2 - base acid, Answer Bank, base, acid
A diprotic weak acid, H2Y, is titrated with a standardized, solution of NaOH. From a graph of the titration, identify what ions, are present in the highest concentration at the second equivalence, ...
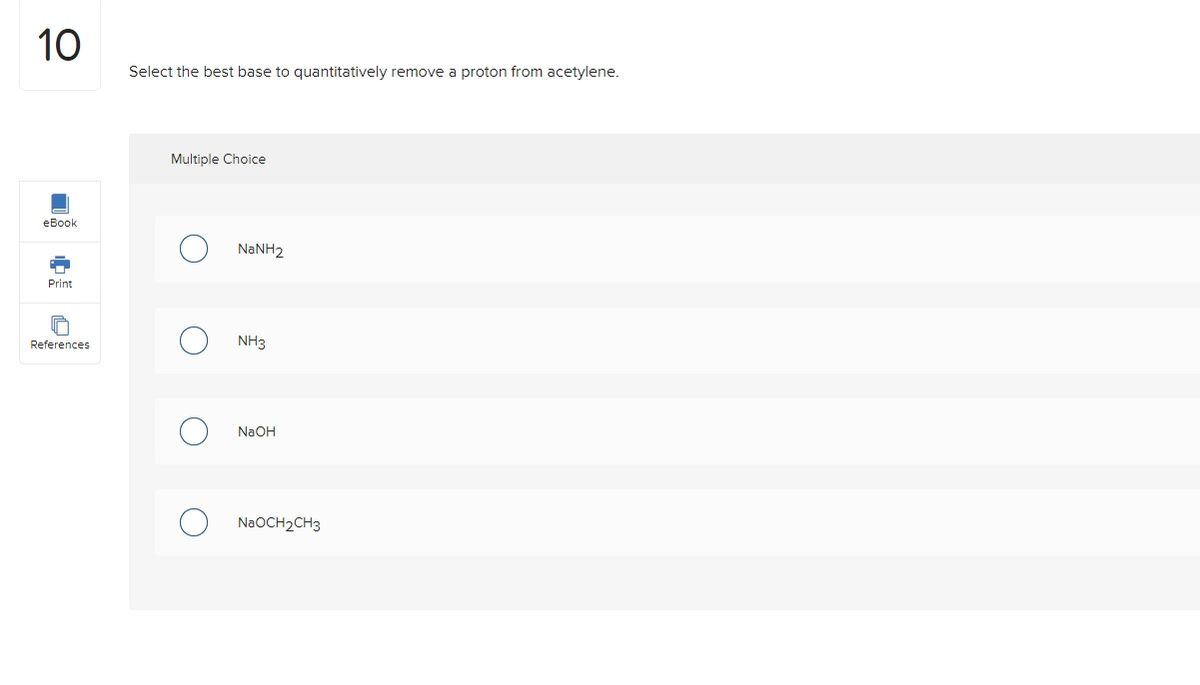



























Post a Comment for "44 h2y acid or base"